The Intersection of Dermatology and Infectious Disease: A Visual Guide
There is a constant demand in public health to address diagnostic decision-making broadly and not focus solely on the diagnosis of the moment. Nearly 20 years ago, health systems and government agencies realized clinicians’ need for accurate and timely information to assist infectious disease diagnosis and surveillance following the threat of bioterrorism triggered by the September 11, 2001 terrorist attacks as well as anthrax spread via the US mail.

Today, clinicians need to recognize COVID-19 as well as all the other diseases that are in the differential diagnoses of fever, cough, abdominal pain, headache, loss of sense of taste or smell, rash of toes, and more. Skin is the window into a person’s overall health. Still, generalists receive an average of 21 hours of training in dermatology,1 making it difficult for them to accurately diagnose skin conditions and their corresponding illnesses.
As dermatologists, we should be empowering all front-line health care clinicians with tools and knowledge that will help them make better decisions in the exam room, whether in person or via telemedicine. Recognizing the cutaneous signs and symptoms of infectious diseases is key in early diagnosis and harm reduction.
The following six infectious diseases have had a significant impact on public health over the past few decades and have placed a spotlight on the importance of dermatology skills in infectious disease diagnosis. Disease information is derived from diagnosis summaries in VisualDx.
Anthrax
Anthrax sent through the mail after the September 11, 2001 terrorist attacks triggered an immediate effort by the US government to prepare for the possibility of bioterrorism broadly. Public health officials sought to rapidly educate medical professionals, in particular emergency physicians and generalists, to quickly recognize signs and symptoms of bioterrorism diagnoses that they were almost universally unfamiliar with.

A depressed, black eschar on the chest.
(All images used with permission from VisualDx.)
Cutaneous anthrax lesions begin as nonpainful, pruritic papules before forming clusters of vesicles to ulcers within one to two days following exposure. The ulcers then develop into depressed, black eschars over the next two to five days. The arms, face, and neck are most commonly affected. Some lesions may have extensive edema.
Without treatment, cutaneous anthrax can progress to a systemic form with a mortality rate of approximately 20 percent.
Measles
Measles is transmitted via respiratory droplets and is highly infectious. The incubation period after the measles virus enters the upper respiratory tract and nasal passages is about 10 days, with the rash typically appearing about 14 days after a person is exposed. Infected individuals are considered contagious from four days before to four days after the rash appears.
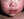
Widespread erythematous patches on the face and chest of a child.
The exanthem consists of erythematous macules and papules beginning at the forehead and behind the ears, eventually spreading down the neck, upper extremities, trunk, and lower extremities. Confluent lesions can occur on the face. The rash typically peaks for three to four days, then begins to fade at day five in the same manner in which it appeared. Desquamation typically occurs after approximately one week. Thrombocytopenia, with resultant purpuric lesions, may complicate measles.
Oral lesions called Koplik spots may develop two to three days after symptoms begin and prior to the generalized cutaneous eruption. Look for minute, white papules, which may have a central bluish-white portion, usually opposite the second molars on the buccal mucosa. They can also appear red. Bluish-gray or white papules can also be seen on the tonsils.
Although measles was declared eliminated in the United States in 2000, outbreaks resulting from imported cases continue to occur. There was a high number of reported measles cases in the United States from January through July 2019, with 1,100 cases from 30 states reported to the Centers for Disease Control and Prevention (CDC), largely from under-immunized communities.
Chikungunya
Chikungunya is transmitted by the bite of infected mosquitoes. The rash typically begins as flushing of the face and trunk and progresses to macules and then erythematous papules on the trunk and extremities. It is associated with painful small joint polyarticular arthralgias. Occasionally, the rash may also appear on the face, palms, and soles. It may be difficult to identify in patients with dark skin.

Erythematous macules on the palms and some papules on the wrist.
Oral manifestations are a less frequent occurrence and are seen more commonly in children. Perineal, genital, and intertriginous ulceration has also been reported.
Variant presentations include the presence of petechiae, purpura, targetoid lesions, vasculitic lesions, or vesicles and bullae. The vesiculobullous form is more commonly seen in children.
Chikungunya was once considered a tropical disease, found mainly in Africa, India, and Southeast Asia. However, outbreaks have occurred in southern Europe, the Caribbean Islands, and Central and South America. In 2014, locally transmitted cases were reported in Florida.
Lyme disease
Lyme disease is the result of infection with the bacteria Borrelia burgdorferi that is transmitted by infected ticks. The tick can be found attached to the skin in many cases.
Erythema migrans, the classic unraised, red “bull’s-eye” lesion on the skin, will appear days to weeks after the bite. However, about 25 percent of affected people never get this lesion. Some may complain of flu-like symptoms, including fever; head, neck, and joint pain; and generalized muscle pain. The lesion will resolve without treatment in about a month.

A large erythematous, annular plaque with a “bull’s-eye” appearance.
In most cases, Lyme disease can be eliminated with antibiotics, especially if treatment is started when symptoms are first noted. Untreated Lyme disease may affect the joints, heart, and nervous system.
Syphilis
Syphilis is considered “the great mimicker” and has stumped even the most seasoned clinicians. According to the CDC, primary and secondary syphilis rates have increased in the United States almost every year since 2001.2
It is important to recognize infection in the primary stage, as lesions in this stage will heal without therapy and all patients who contract syphilis will go on to develop secondary syphilis if the initial infection is not treated. When treated early enough, syphilis can be completely cured.
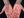
Untreated syphilis that progressed to secondary syphilis, seen here as discrete orange-red papules, some with thick overlying scale, on the palms.
Look for one or more painless ulcers (chancres) ranging from a few millimeters to several centimeters at the site of inoculation. The classically described round, punched-out ulcer is pink and shiny and has a nonpurulent, clean base with scant serous exudate and an indurated raised border. Genital sites affected in women are the cervix, vagina, vulva, and clitoris. In men, the chancre is easily seen on the penis.
There may be a mixed infection of syphilis with any other sexually transmitted disease (STD) that may change the appearance of the ulcer. Atypical chancres have been noted to present as painful erosions or ulcers with soft or hard borders with a dirty or clean base. Anal erosions, ulcers, and fissures may also be seen.
Secondary syphilis typically occurs one to three months after the first stage of syphilis. It has a wide range of cutaneous manifestations. These include a generalized nonpruritic papulosquamous eruption including the palms and soles, with pink, violaceous, or copper-colored papules, each with a collarette of scale. Patchy alopecia of the scalp is also observed in secondary syphilis. Ocular symptoms may include lacrimation, photophobia, and red, painful eyes.
MRSA
Skin and soft tissue infections including cellulitis, abscesses, impetigo, folliculitis, and furunculosis can all be caused by infection with Staphylococcus aureus, including methicillin-resistant S aureus (MRSA). Over the past two decades, the incidence of community-associated MRSA (CA-MRSA) has been increasing. In many parts of the United States, more than 50 percent of community-associated S aureus isolates are MRSA. Hence, infections where S aureus is suspected should be considered MRSA unless susceptibility studies demonstrate otherwise.

Culture-confirmed MRSA showing a furuncle with overlying scale and central purulence on the knee.
The most common manifestations of CA-MRSA infections are skin and subcutaneous infections. MRSA skin infections present similarly to any bacterial skin infection, although skin abscesses caused by MRSA can often appear larger and more severe than other infections. Look for follicular pustules, furuncles (boils), abscesses, and bullous and non-bullous impetigo.
Empowering Physicians at the Point of Care
As a dermatologist and health informaticist, one of my missions is to put knowledge and visualization at the point of care to drive better treatment, testing, and diagnostic decisions. While the COVID-19 pandemic is placing additional pressure on health care professionals, the use of tools that provide education in the skin exam, improve diagnostic accuracy, and speed medical care is essential. Clinical decision support can be used as a second opinion right at the point of care, giving clinicians the information they need to make informed decisions and improve patient engagement.
As we wind down from the acute phase of this current pandemic, we should consider the structure and functionality of a modern public health digital infrastructure. Since no one knows which public health threats will emerge in the future as our next crisis, the only sensible strategy is to prepare for all infectious diseases and diseases of public health significance. We must make sure we are empowering all clinicians with the tools and knowledge to recognize early skin manifestations of infectious diseases.
1. Fleischer AB Jr, Herbert CR, Feldman SR, O’Brien F. Diagnosis of skin disease by nondermatologists. Am J Manag Care. 2000 Oct;6(10):1149-56.
2. Sexually Transmitted Disease Surveillance 2017. Centers for Disease Control and Prevention. Accessed at: https://www.cdc.gov/std/stats17/2017-STD-Surveillance-Report_CDC-clearance-9.10.18.pdf.
Ready to Claim Your Credits?
You have attempts to pass this post-test. Take your time and review carefully before submitting.
Good luck!
Recommended
- Infections & Infectious Disease
Systemic Corticosteroids, Chronic Inflammatory Skin Disease, and COVID-19: What Do We Know?





